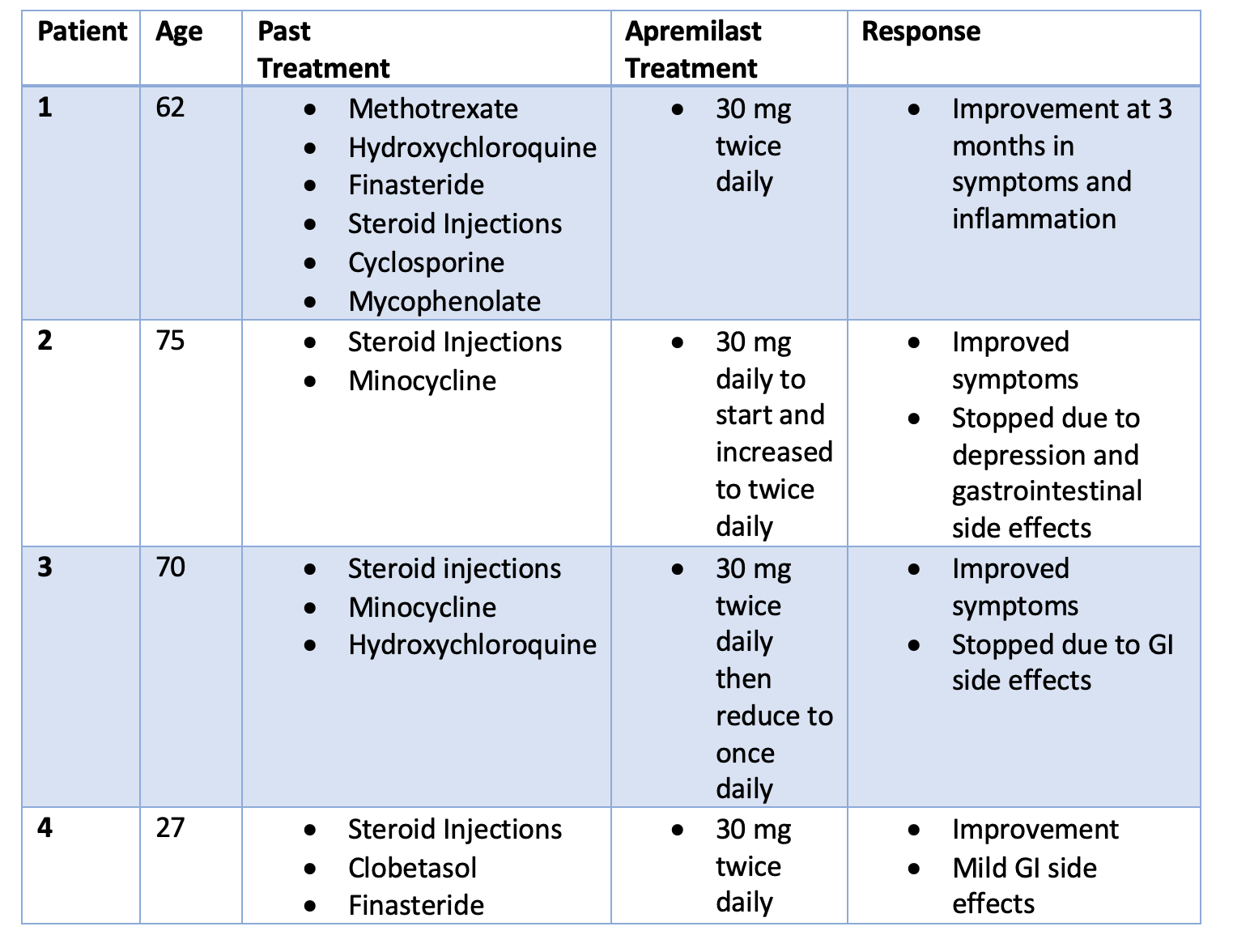
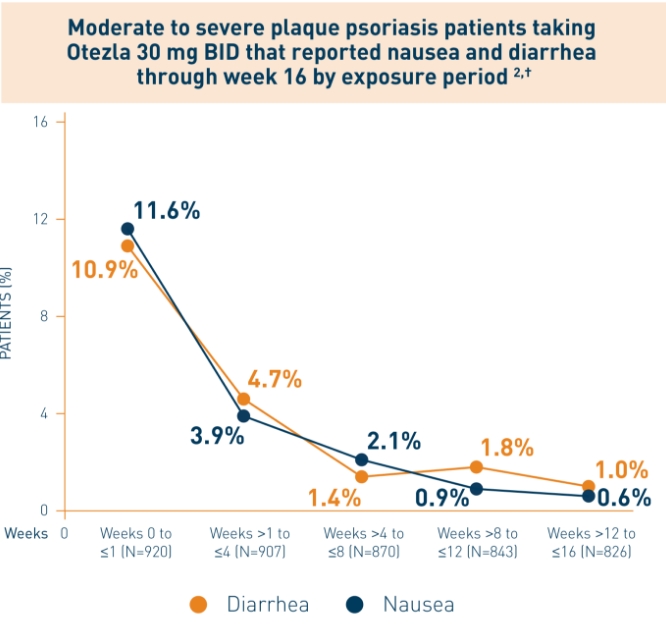
These highlights do not include all the information needed to use OTEZLA safely and effectively. See full prescribing information for OTEZLA. OTEZLA® (apremilast) tablets, for oral use Initial U.S. approval: 2014

The efficacy and safety of apremilast, etanercept and placebo in patients with moderate‐to‐severe plaque psoriasis: 52‐week results from a phase IIIb, randomized, placebo‐controlled trial (LIBERATE) - Reich - 2017 - Journal of
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use OTEZLA safely and effec

Efficacy and safety of apremilast in patients with moderate‐to‐severe plaque psoriasis of the scalp: results up to 32 weeks from a randomized, phase III study - Van Voorhees - 2021 - British

The FDA-approved drug apremilast suppresses alcohol intake: clinical and pre-clinical validation | bioRxiv