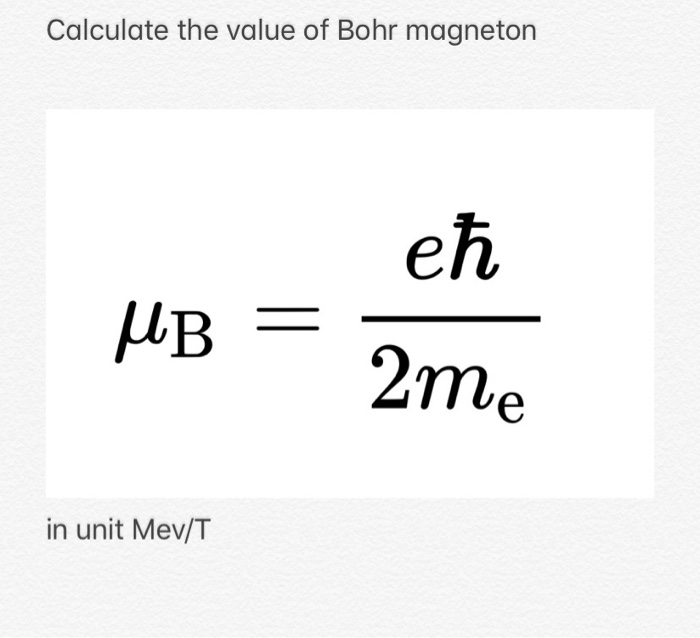![The \"spin-only\" magnetic moment [in units of Bohr magneton, `(mu_B)`] or `Ni^(2+)` in aquenous - YouTube The \"spin-only\" magnetic moment [in units of Bohr magneton, `(mu_B)`] or `Ni^(2+)` in aquenous - YouTube](https://i.ytimg.com/vi/4jAu-kiL0G0/maxresdefault.jpg)
The \"spin-only\" magnetic moment [in units of Bohr magneton, `(mu_B)`] or `Ni^(2+)` in aquenous - YouTube
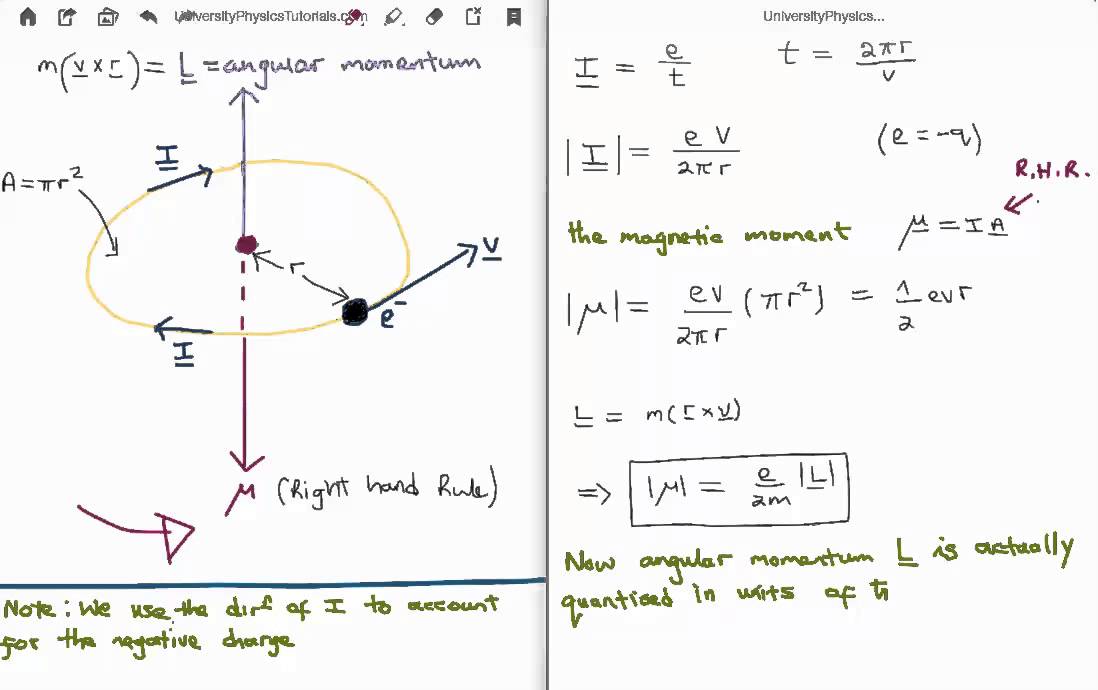
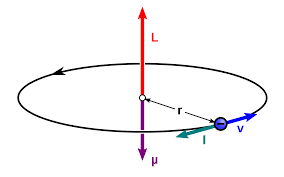
What Is Bohr Magneton? Deduce The Expression Of Bohr Magneton And Find Out Its Numerical Value. What Do You Mean By Nuclear Magnetic Resonance. Write Down The Applications Nuclear Magnetic Resonance. -

What is the magnetic field, created by ion of Fe^{+2} (for electron of conductivity), if the ion magnetic moment equals 5.1 Bohr's magneton?
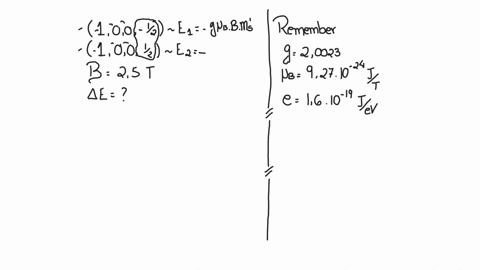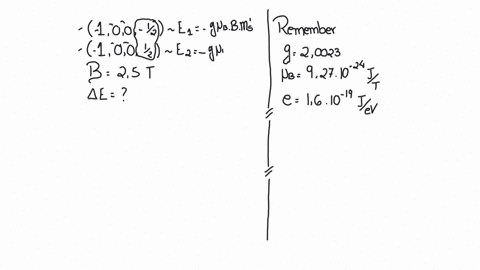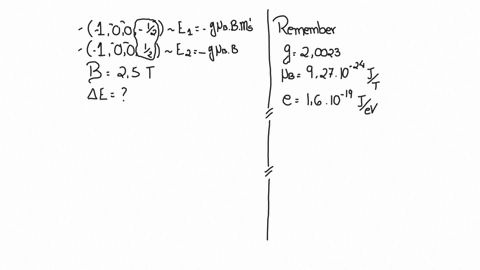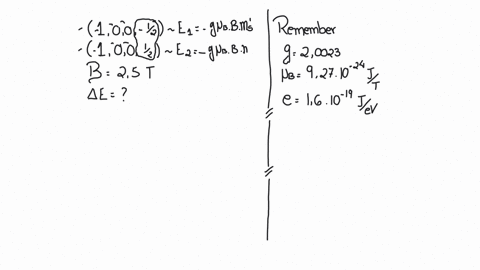
SOLVED:(I) Verify that the Bohr magneton has the value \mu_{B}=9.27 \times 10^{-24} \mathrm{J} / \mathrm{T} (see Eq. 12) . \mu_{\mathrm{B}}=\frac{e \hbar}{2 m}

What is the correct value of Bohr magneton? | 12 | MOVING CHARGES AND MAGNETISM | PHYSICS | NCE... - YouTube

The value of the magnetic moment of a particular ion is `2.83` Bohr magneton. The ion is :- - YouTube