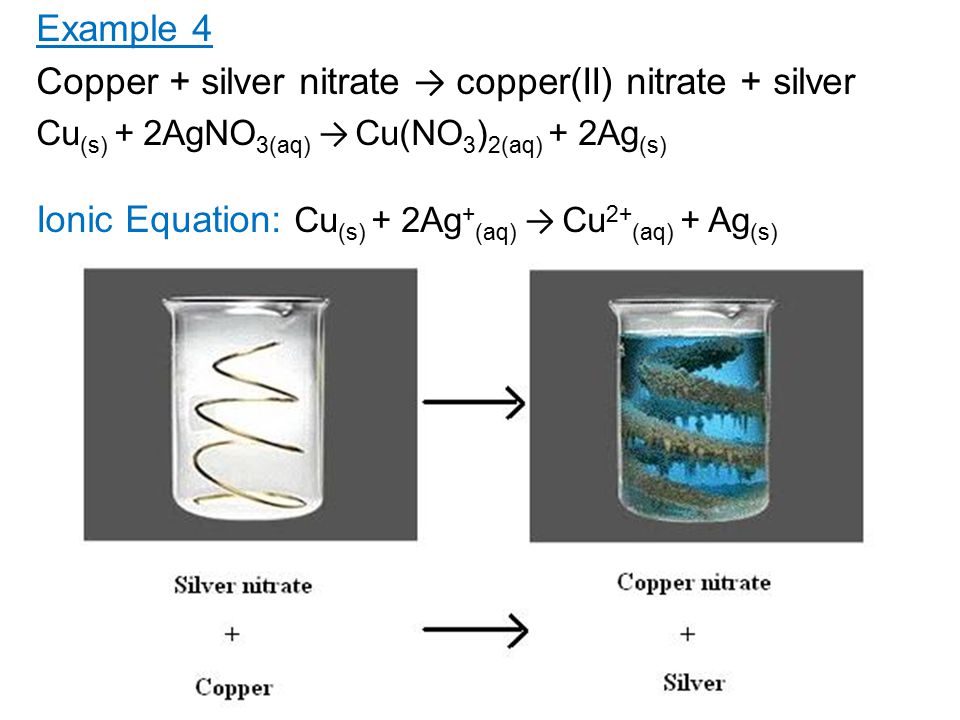
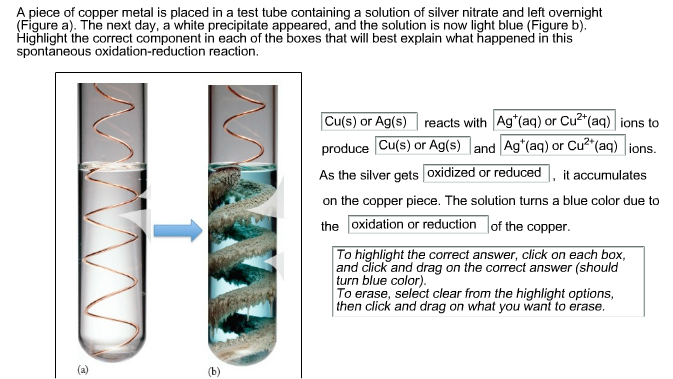
When copper is dipped in the solution of silver nitrate, the solution turns blue. Give the reason along with chemical equation?

STOCK IMAGE, , JB7956, 01B466TS , Science Source - Search Medical & Scientific Stock Photos at MedicalImages.com

SOLVED: The reaction between solid copper and aqueous silver nitrate produces solid silver and blue solution, as represented by the balanced equation shown below: Cu(s) 2AgNO (aq) Cu(NO )(aq) 2Ag(s) Based on
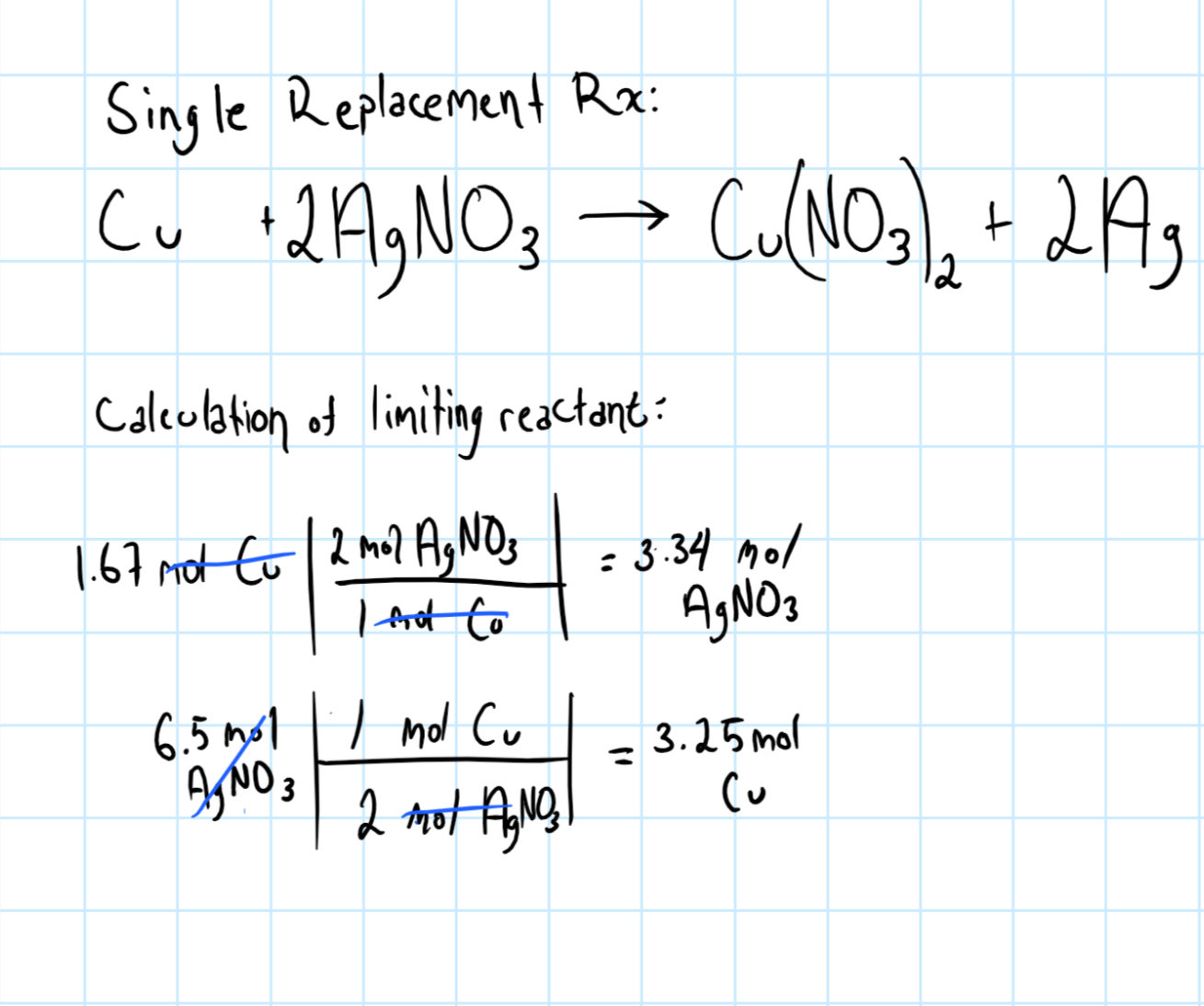
/copper-wire-immersed-in-silver-nitrate-causing-blue-colour-81991997-582f14595f9b58d5b1a9b484.jpg)